Antibody industrial park
The Antibody Industrial Park has completed the construction and 2 sets of 3500L stainless steel bioreactors and their supporting downstream purification production lines, 3 sets of 500L disposable reactors and their supporting downstream purification production lines, 1 set of 200L antibody drug coupling reactor and its supporting isolator system and purification production line, and a vial of aseptic injection solution washing, drying, filling and sealing linkage production line are put into use . There are also supporting facilities and plants such as sewage treatment station, material warehouse, dangerous goods warehouse, liquid oxygen station, etc. built in the park. To date, we are continuing to expand our monoclonal antibody production capacity to meet the demand for industrialisation of a number of monoclonal antibodies in our research and development pipeline that are currently in the early clinical or research and development stage. Our antibody industrial park can produce monoclonal antibody drugs and ADC drugs at different stages, including clinical use and commercial production, from stock solutions to sterile formulation production.

Quality Management System
Based on the ICHQ10 quality system model, we have established a systematic, rigorous and perfect quality management system with the minimum standards of the major official GMP requirements of the United States, the European Union, and China, covering the entire life cycle of the product: from the research and development stage of the drug , technology transfer, commercial production to product delisting.
We take "observing laws and regulations, emphasizing reputation, ensuring production behavior norms; grasping the foundation, strict management, and continuously improving the quality system" as our quality policy, and "establishing, ensuring that the quality of drugs meets registration requirements and quality standards; improving the quality awareness of production personnel and post operation skills, implement scientific and advanced technology" as the quality goal at this stage, make full use of knowledge management and quality risk management tools, and establish a complete file system to guide production, inspection, plant equipment maintenance, quality management, etc. development of the work.
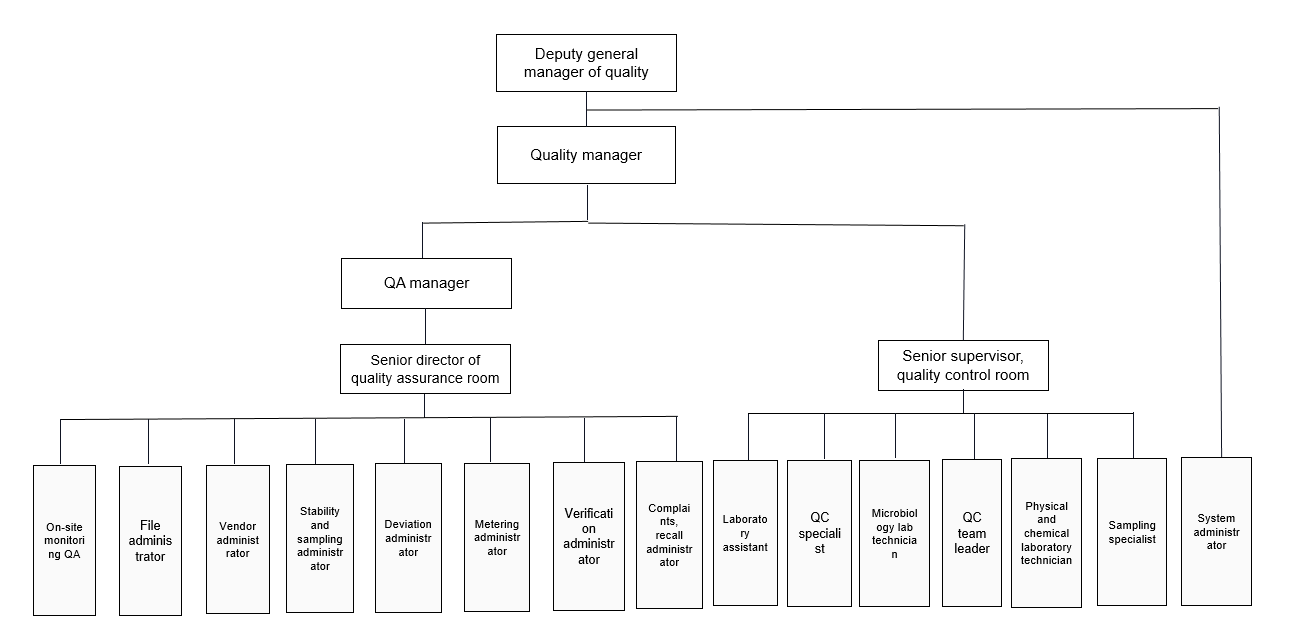
The company attaches importance to product quality and safety, the establishment of the Quality Department, divided into two secondary departments of the Quality Assurance Office and the Quality Inspection Office, has been adhering to the quality management concept of "one dose of medicine, two lives", according to the requirements of the GMP management standards, to achieve "source control, process control, product release, after-sales service" full life-cycle management to ensure product quality. According to the requirements of GMP management standards, we achieve the whole life cycle management of "source control, process control, product release and after-sales service" to ensure product quality and achieve continuous improvement and enhancement of quality management level.
In addition, we have established a quality management team with responsibility, execution and a high sense of quality, and the staffing reflects our strong commitment to the quality management function. In order to ensure stable production processes and reliable inspection data, a large amount of confirmation and validation work has been completed. We attach great importance to the management of data reliability and have established a programme document for data reliability management. The paper and electronic data of pharmaceutical products in R&D, production and inspection comply with the ALCOA principles: traceability, clarity, synchronization, originality and accuracy. Our pursuit of quality drives us to maintain a high level of responsibility in everything we do.